 
The proposed algorithm is able to cope with general molecular rearrangement reactions occurring during EI in GC/MS measurements. Here, we present the fragment formula calculator (FFC) algorithm that can calculate chemical formulas for fragment ions where the chemical bonding (e.g., Lewis structures) of the intact molecule is known. For that reason, we developed an algorithm, based on a systematic bond cleavage, to determine chemical formulas and retained atoms for EI derived fragment ions. However, the process of mass spectral fragmentation is complex, and identifying the substructures and chemical formulas for these fragment ions is nontrivial. Fragment ions containing different carbon atoms are of special interest, since they can carry different flux information. For example, 13C-metabolic flux analyses ( 13C-MFA) are dependent on the exact knowledge of the number and position of retained carbon atoms of the unfragmented molecule. Moreover, the positional distribution of stable isotopes within a fragment ion is of high interest for stable isotope-assisted metabolomics techniques. Hence, the correct assignment of chemical formulas to fragment ions is of crucial importance for correct MIDs. The chemical formula of a fragment ion is usually applied to derive the correction matrix for accurate MID calculation. The analysis of fragment ions formed during EI, which contain only specific parts of the original molecule can provide valuable information on the positional distribution of the label. Most commonly, MIDs are derived from gas chromatography/electron ionization mass spectrometry (GC/EI-MS) measurements. The empirical formula mass of a covalent compound may be compared to the compound’s molecular or molar mass to derive a molecular formula.The accurate determination of mass isotopomer distributions (MID) is of great significance for stable isotope-labeling experiments. A compound’s percent composition provides the mass percentage of each element in the compound, and it is often experimentally determined and used to derive the compound’s empirical formula. The chemical identity of a substance is defined by the types and relative numbers of atoms composing its fundamental entities (molecules in the case of covalent compounds, ions in the case of ionic compounds). The molar mass is approximately 194 g/mol. Molar mass of molecular formula / Molar mass empirical formula = 90.3 g/mol / 46.0055 g/mol = 1.963 (to 3 SF) = 2Ĭaffeine contains hydrogen, carbon, nitrogen and oxygen. Then, we need to use the molar mass values to determine the molecular formula:Įmpirical formula molar mass = 14.0067 g/mol + 2(15.9994 g/mol) = 46.0055 g/mol to 2 sig fig’s) is close enough to round to the whole number, or NO 2 The corresponding numbers of atoms (in moles) are: Consider a sample of compound determined to contain 1.71 g C and 0.287 g H. We then consider the moles of each element relative to each other, converting these numbers into a whole-number ratio that can be used to derive the empirical formula of the substance. To accomplish this, we can use molar masses to convert the mass of each element to a number of moles. 
Therefore, any experimentally derived data involving mass must be used to derive the corresponding numbers of atoms in the compound. 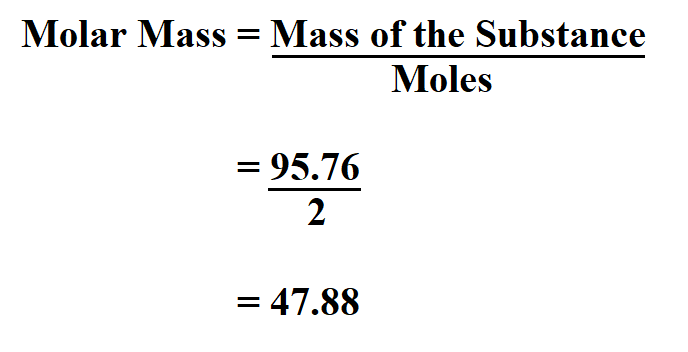
However, we must keep in mind that chemical formulas represent the relative numbers, not masses, of atoms in the substance. Thus, the empirical formula = C 12/2H 4/2Cl 4/2O 2/2= C 6H 2Cl 2Oĭetermine the empirical formula for the following compounds:Ī) C 3H 8N b) CCl 4 c) C 2H 5 Determination of Empirical FormulasĪs previously mentioned, the most common approach to determining a compound’s chemical formula is to first measure the masses of its constituent elements. Determine the empirical formula for dioxin (C 12H 4Cl 4O 2), a very powerful poison.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
